|
The only reported nonhematologic grade ≥3 adverse events that occurred more frequently in the radium-223–treated patient group were anorexia (2% versus 1%) and a decreased appetite (2 patients vs. In general, radium-223 was well tolerated by patients, with grade 3 or 4 adverse events occurring more frequently in the placebo-treated group (62%) than in patients treated with radium-223 (56%). The median overall survival was 65 weeks in the radium-223–treated group, and 46 weeks in the placebo-treated group ( P = 0.056). In a follow-up report, 24 months after the first injection of study medication, no long-term treatment-related toxicity was noted ( 24). Severe nonhematologic adverse events occurred in 3 (9.1%) radium-223–treated patients and in 5 (16.7%) placebo-treated patients. Grades 3 and 4 adverse events occurred in 3 (9.1%) and 0 (0%) radium-223–treated patients and in 1 (3.3%) and 1 (3.3%) placebo-treated patients, respectively. None of the 33 radium-223-treated patients discontinued treatment because of adverse events. Low toxicity of radium-223 was confirmed in this phase II study. The median time to skeletal-related event was 14 weeks in the radium-223–treated group versus 11 weeks in patients treated with placebo ( P = 0.257 ref. Therefore, the half-lives of radon-219 (4.0 seconds), bismuth-211 (2.1 minutes), and thallium-207 (4.8 minutes) seem to be long enough to allow diffusion from the primary accumulation site.Ī subsequent phase II study compared treatment of 50 kBq/kg radium-223 with placebo treatment in 64 CRPC patients with painful bone metastases ( 23, 24). These daughter nuclides do not have a chemical similarity to calcium ions. The half-life of radium-223 is 11.4 days the half-lives of its daughter nuclides range from seconds to minutes. Because of the size and high energy of α-particles, these particles are highly effective in inducing double-strand breaks in DNA within 100 μm. Both α- and β-irradiation can induce local therapy by inducing damage in the surrounding tissue. During the decay of each radium-223 isotope, 4 α-particles and 2 electrons (β-particles) are emitted ( Table 1). Radium-223 is an α-emitting radioisotope that decays via 7 daughter nuclides before it stabilizes as lead-207 ( Fig. 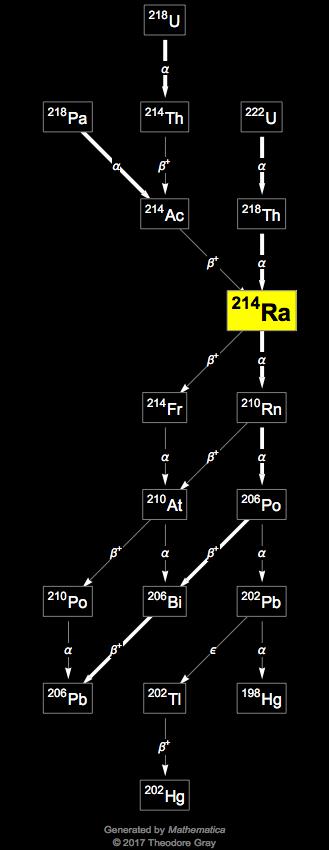
In ionic form, radium accumulates in bones at areas with increased bone turnover because of its chemical similarity to calcium ions both are alkaline earth metals ( 18, 19). Moreover, as surrounding tissue, including the bone marrow, can be damaged as well, significant adverse events such as bone marrow failure may occur in treated patients.Ģ23RaCl 2 is a water-soluble radium salt. However, the duration of response is limited the effect of these treatments on overall survival has not been studied. Such therapies result in symptomatic relief in more than half of patients ( 16, 17). Multiple agents have been approved by the US Food and Drug Administration (FDA) for the palliative treatment of bone metastases in mCRPC patients, such as external beam radiotherapy and the β-emitting radiopharmaceuticals strontium-89 (1.5–2.2 MBq/kg) and samarium-153 (37 MBq/kg Table 1). Hence, specific treatment of bone metastases may significantly lower the burden of prostate cancer disease ( 14, 15). Bone metastases may lead to severe morbidity, such as bone marrow failure, pathological fractures, and spinal cord compression, reducing quality of life and potentially resulting in death ( 12, 13). Prostate cancer primarily metastasizes to the bone ( 11). Further studies are required to optimize its dosing and to confirm its efficacy and safety in cancer patients. 223RaCl 2 is the first bone-targeting antitumor therapy that received FDA approval based on a significant extended median overall survival. The radioisotope was well tolerated and gave limited bone marrow suppression. At a prespecified interim analysis, the primary endpoint of median overall survival was significantly extended by 3.6 months in patients treated with radium-223 compared with placebo ( P < 0.001). In a phase III study (ALSYMPCA), mCRPC patients with bone metastases received best standard-of-care treatment with placebo or 223RaCl 2. 
Radium-223 is an α-emitting alkaline earth metal ion, which, similar to calcium ions, accumulates in the bone. Food and Drug Administration (FDA) approved radium-223 chloride ( 223RaCl 2) for the treatment of mCRPC patients whose metastases are limited to the bones. The treatment scope for patients with metastatic castrate-resistant prostate cancer (mCRPC) is rapidly expanding.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
